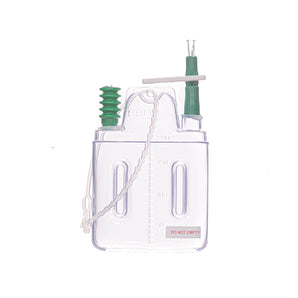
Dynadress
Transparent IV Fixation Dressing with Chlorhexidine Gluconate Pad (CHG)
Transparent IV Fixation Dressing with Chlorhexidine Gluconate Pad (CHG)
CHG IV Dressing: The Clinical Standard for Antimicrobial Protection and Vascular Site Securement
CHG IV Dressing is a specialised medical device engineered to provide a sterile barrier and continuous antimicrobial protection at the insertion site of intravenous catheters. In the South African healthcare landscape—where the prevalence of Hospital-Acquired Infections (HAIs) and antibiotic-resistant pathogens is a significant clinical challenge—the use of a high-performance chg dressing is no longer a luxury but a necessity for patient safety. Dynamed Pharmaceuticals, a youth-driven manufacturer based in Durban, delivers an elite IV Fixation Dressing that integrates a Chlorhexidine Gluconate (CHG) pad with a highly breathable transparent film. This combination ensures that the catheter site remains visible, secure, and protected against microbial colonization for the duration of the catheter’s wear time.
For clinical directors, nursing managers, and hospital procurement officers, selecting an Antimicrobial IV Securement solution involves balancing efficacy, regulatory compliance, and cost-efficiency. Dynamed’s CHG IV Dressing is manufactured in an ISO 8 cleanroom and is fully SAHPRA-compliant, offering a reliable local alternative to expensive international imports without compromising on clinical outcomes. By utilizing IV Fixation with CHG Pad technology, medical facilities can significantly reduce the risk of Catheter-Related Bloodstream Infections (CRBSIs), which are a leading cause of morbidity and increased healthcare costs in both private and public sectors.
The Science of Antimicrobial Protection in Vascular Access
The core of the CHG IV Dressing is the integrated medical-grade pad impregnated with Chlorhexidine Gluconate. Chlorhexidine is a potent antiseptic with a broad spectrum of activity against Gram-positive and Gram-negative bacteria, as well as yeast. Unlike liquid antiseptics applied during skin prep, which may lose efficacy shortly after application, the chg dressing provides a sustained release of antimicrobial agents directly onto the skin surrounding the insertion site.
This continuous antimicrobial protection is critical because skin flora, such as Staphylococcus aureus and Staphylococcus epidermidis, are the primary sources of catheter contamination. When a Transparent CHG Film Dressing is applied, the CHG pad suppresses the regrowth of these micro-organisms for up to seven days. This matches the standard wear-time protocols recommended by the Centers for Disease Control and Prevention (CDC), reducing the frequency of dressing changes and minimising the opportunities for pathogens to enter the bloodstream.
The Mechanism of IV Fixation with CHG Pad
An IV Fixation with CHG Pad functions through a dual-action mechanism. First, the adhesive polyurethane film provides the mechanical securement necessary to prevent catheter dislodgement. Second, the CHG-impregnated pad creates a zone of inhibition around the puncture site. This Antimicrobial IV Securement is particularly vital for Central Venous Catheters (CVCs) and Arterial Lines, where the consequences of an infection are most severe.
Furthermore, Dynamed’s Transparent CHG Film Dressing is designed with a high Moisture Vapor Transmission Rate (MVTR). This ensures that moisture and perspiration are wicked away from the skin, preventing maceration and ensuring that the adhesive maintains its integrity. A dry environment under the chg dressing is essential for both skin health and long-term securement, especially in South Africa’s varied climatic conditions.
Clinical Benefits for Nursing and Patient Care
For nursing professionals, the efficiency of an IV Fixation Dressing directly impacts daily clinical workflows. The CHG IV Dressing is designed for intuitive application, featuring a reinforced border and securement strips that allow for a one-handed technique. This speed is crucial in emergency services and high-pressure ICU environments where every second counts.
Visibility and Site Monitoring
The transparency of the Transparent CHG Film Dressing is a vital safety feature. It allows clinicians to monitor the insertion site for signs of phlebitis, infiltration, or infection without removing the dressing. Unnecessary dressing changes are a known risk factor for catheter contamination; therefore, a chg dressing that remains in place while providing a clear view of the site is a significant clinical advantage.
By providing antimicrobial protection and visibility, the IV Fixation Dressing allows nursing teams to adhere to "best practice" monitoring protocols. If redness or swelling is observed through the Transparent CHG Film Dressing, immediate intervention can be taken, potentially saving the patient from a systemic infection and the hospital from the costs of extended care.
Procurement Excellence for South African Hospitals
The ideal audience for Dynamed’s medical consumables - hospital procurement managers and clinical directors - prioritizes quality and regulatory compliance. In South Africa, the medical supply chain is often strained by a reliance on imported products that are subject to currency fluctuations and long lead times. Dynamed Pharmaceuticals addresses this by being a youth-driven, local manufacturer of the CHG IV Dressing.
Compliance with International Standards
Dynamed’s commitment to elite manufacturing is evidenced by our ISO 13485 certification and our GMP-aligned production processes. Every chg dressing is produced under stringent quality control, ensuring that each batch is sterile and effective. This dedication to quality makes our Antimicrobial IV Securement solutions a preferred choice for public-sector tender committees and private-sector pharmacy buyers.
When a facility selects IV Fixation with CHG Pad from Dynamed, they are not just buying a commodity; they are partnering with a company that understands the clinical environment. Our IV Fixation Dressing simplifies procurement approvals because it carries the necessary credentials to pass rigorous clinical audits and safety inspections.
Reducing the Economic Burden of CRBSI with CHG Dressing
Catheter-Related Bloodstream Infections (CRBSIs) are not only a patient safety issue but also a significant financial burden on the South African healthcare system. A single case of CRBSI can extend a patient’s hospital stay by several weeks and cost the facility hundreds of thousands of Rands in additional medications, staffing, and diagnostics. The proactive use of CHG IV Dressing technology is a cost-effective insurance policy.
Studies have shown that antimicrobial protection provided by CHG-impregnated dressings can reduce CRBSI rates by over 60% compared to standard transparent dressings. By investing in Antimicrobial IV Securement, hospital administrators can realize a high return on investment (ROI) through lower infection rates and improved patient throughput. Dynamed’s IV Fixation with CHG Pad offers this elite level of protection at a price point that makes it accessible for large-scale public sector rollouts and NGOs.
Comparison: Traditional vs. CHG-Enhanced IV Fixation
To understand the value of the CHG IV Dressing, it is helpful to compare it to traditional securement methods:
|
Feature |
Standard Transparent Dressing |
CHG IV Dressing |
|
Microbial Barrier |
Physical barrier only |
Physical + Continuous antimicrobial protection |
|
Infection Reduction |
Baseline |
Reduced CRBSI risk by 60%+ |
|
Site Visibility |
High |
High (with integrated pad) |
|
Wear Time |
Up to 7 days (if clean) |
Up to 7 days (with sustained CHG release) |
|
Best Use |
Simple peripheral IVs |
CVCs, PICCs, Arterial Lines, High-risk PIVCs |
While a standard IV Fixation Dressing is suitable for low-risk, short-term access, any patient requiring a central line or long-term catheterisation should be managed with a chg dressing. The addition of the CHG pad transforms the securement device into an active therapeutic tool, providing Antimicrobial IV Securement that passively protects the patient 24 hours a day.
Technical Specifications for Dynamed’s Transparent CHG Film Dressing
Dynamed’s Transparent CHG Film Dressing is designed with precision to meet the needs of South African medical officers. Key technical features include:
- CHG Concentration: The integrated pad contains a standardised concentration of Chlorhexidine Gluconate designed for effective bacterial suppression without causing skin irritation in most patients.
- Polyurethane Film: A high-MVTR film that allows for breathability while remaining waterproof and impermeable to external bacteria.
- Adhesive System: A hypoallergenic, medical-grade acrylic adhesive that provides IV Fixation Dressing stability even on diaphoretic (sweating) patients.
- Sterilization: Every CHG IV Dressing is sterilized using Ethylene Oxide (EO) or Gamma Irradiation, ensuring a shelf-life suitable for long-term hospital inventory.
The Durban Manufacturing Advantage
Being a "medical consumables manufacturer Durban," Dynamed offers a strategic advantage to the SADC region. By manufacturing our IV Fixation with CHG Pad locally, we provide stable lead times and predictable stock availability. This is a critical factor for hospital administrators who cannot afford stock-outs of essential Antimicrobial IV Securement products. Our youth-driven team is responsive and committed to innovation, ensuring that Dynamed stays at the forefront of wound care technology.
Step-by-Step Application Guide for CHG IV Dressing
To maximise the antimicrobial protection provided by the chg dressing, correct application is essential:
-
Preparation: Perform hand hygiene and clean the skin around the insertion site using a 2% CHG/70% Alcohol prep. Allow the skin to dry completely to ensure the IV Fixation Dressing adheres correctly.
-
Positioning: Remove the backing of the CHG IV Dressing. Position the CHG pad directly over the catheter insertion site.
-
Smooth Down: Press the Transparent CHG Film Dressing onto the skin, smoothing from the center outward to eliminate bubbles.
-
Securement: Use the included securement strips to anchor the catheter hub. This prevents "pistoning" (movement of the catheter in and out of the vein), which is a common cause of infection.
-
Documentation: Label the chg dressing with the date of application.
Regulatory Compliance: ISO and SAHPRA
For professional healthcare decision-makers, compliance is a non-negotiable metric. Dynamed Pharmaceuticals prioritizes transparency in our manufacturing processes. Our CHG IV Dressing is manufactured in a facility that adheres to ISO 13485 standards, ensuring that our medical devices are consistent and safe for clinical use.
In South Africa, SAHPRA (South African Health Products Regulatory Authority) compliance is the gatekeeper for patient safety. Our IV Fixation with CHG Pad is registered and approved, providing reassurance to public-sector tender committees that they are procuring high-quality, dependable medical consumables. This compliance simplifies the procurement process for hospital administrators, reducing risk and demonstrating consistency at scale.
Why Dynamed is the Preferred Partner for Wound Care
Dynamed Pharmaceuticals is more than just a supplier; we are a partner in improving patient outcomes. Our audience—nurses, clinicians, and medical officers—experience the realities of patient care daily. They are drawn to our CHG IV Dressing because it is designed for efficiency and safety in high-pressure routines.
What matters to us is how the product performs in real clinical scenarios. Whether it is reducing infection risk in a neonatal ward or ensuring Antimicrobial IV Securement during a multi-hour surgery, Dynamed’s chg dressing provides the reliability that healthcare professionals demand. We value transparent communication and are committed to supporting our partners during audits and documentation reviews.
Conclusion: Elevating Standards with Dynamed
In conclusion, the CHG IV Dressing is a vital tool in the modern medical arsenal against vascular access infections. By providing continuous antimicrobial protection, secure IV Fixation Dressing properties, and a clear view for monitoring, it fulfills the highest expectations of clinical excellence.
Dynamed Pharmaceuticals is proud to offer our locally manufactured, SAHPRA-compliant IV Fixation with CHG Pad to the South African and SADC markets. We speak directly to the needs of professionals who refuse to sacrifice safety for cost. With Dynamed, you get the best of both: elite manufacturing standards and accessible pricing. Trust us to be your partner in improving patient comfort, operational efficiency, and clinical performance through superior Antimicrobial IV Securement.
For more information on our wound care solutions or to request a product sample, contact Dynamed today. Let us help you set a new standard for patient safety in your facility.
Frequently Asked Questions (FAQ)
1. How long can a CHG IV Dressing be left in place?
According to international guidelines and manufacturer specifications, a chg dressing can typically be left in place for up to 7 days, provided the site remains clean, dry, and the dressing is intact.
2. Is the Transparent CHG Film Dressing waterproof?
Yes, the polyurethane film used in Dynamed’s dressing is waterproof, allowing patients to shower while protecting the catheter site from external water and bacteria.
3. Can IV Fixation with CHG Pad be used on neonates?
Special caution should be used with CHG products on neonates under 2 months of age due to potential skin sensitivity and risk of systemic absorption. Always follow hospital-specific pediatric protocols.
4. How does Antimicrobial IV Securement reduce costs?
By preventing even a single CRBSI, a hospital can save significantly on the costs of extended stays, expensive antibiotics, and additional nursing care, making the IV Fixation Dressing a highly cost-effective choice.
5. Is Dynamed an ISO-certified manufacturer?
Yes, Dynamed is an ISO 13485-certified medical device manufacturer based in Durban, South Africa, specialising in high-quality, compliant medical consumables.
The Transparent IV Fixation Dressing with Chlorhexidine Gluconate (CHG) Pad is an advanced solution for securing intravenous catheters while providing continuous antimicrobial protection. Designed with a 2% CHG gel pad, it offers immediate and sustained activity against a broad spectrum of microorganisms, including those associated with catheter-related bloodstream infections (CRBSIs).
Have feedback? Give us direct feedback here.